R&D
研究开发
Product Pipeline
- Categories:R&D
- Time of issue:2020-03-12 00:00:00
- Views:0
Description:
Information
After years of research and development, we have a balanced and diversified product pipeline with a strategic focus on dermatology and major cancers.
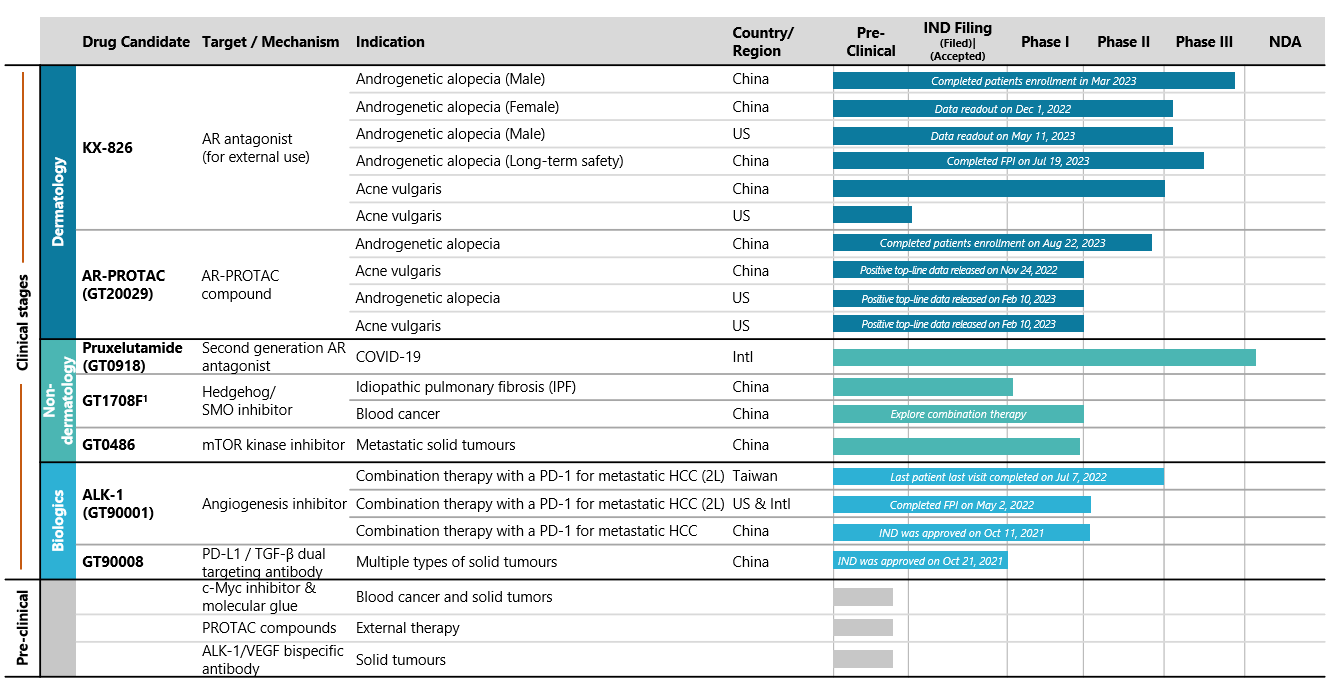
Note: 1.In addition, IND for basal-cell carcinoma indication has been cleared by FDA in the U.S.
Scan the QR code to read on your phone
Business
News Center
Contact Us
Find Us


Copyright: Kintor Pharmaceutical Limited retains all copyrights. 苏ICP备19028699号-2 Powered by 300.cn
Complaints and Suggestions Tel: +86 512 62639935 Email: public@kintor.com.cn




